|
9/4/2023 0 Comments Copper transition metal 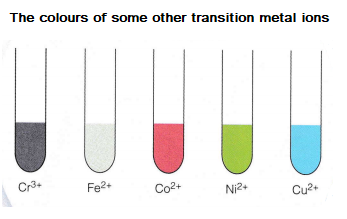
Only 4000 - 5000 µg of Copper (Cu) is required daily in normal diet for an adult. Excess of copper leads to damage the antioxidant enzyme function, oxidative modification of DNA and proteins, lipid oxidation, activate the redox-sensitive genes, suppress the Zinc consumption in the body and also makes anemic by interfering with iron transport. The Copper (Cu) is essential to life, it is the third most abundant trace element in the body after zinc and iron, but whenever the amount 80,000 µg to 100,000 µg of copper intakes by an adult human body from the outsource, becomes toxic. Moist air affects Copper (Cu) and makes green coating of CuSO 4∙3Cu (OH) 2, it is a basic sulphate, which is dissolved in concentrated acids. 
Copper (Cu) occupies d 9-state in various complexes. Copper (Cu) may be Cu 1+ or Cu 2+ in ionic state and known as -ous and -ic suffix, forms Complexes with suitable ions and radicals. Its important ores are Malachite, Chalcocite or Copper glance (Cu 2S), Copper Pyrites (Cu FeS 2) (Cu 2S∙Fe 2S 2). There are three important oxidation states of copper (Cu) that are zero (0) (pure Metal), +1 ous-suffix added compounds, +2 ic-suffix added compounds. Mainly it is found in America at Superior, Ural Mountain in Siberia, Assam and (Singhbhum), Jharkhand State of India. Its electronic configuration is 3d 104s 1. It is a member of Group-1B in periodic table with Silver (Ag) and Gold (Au). 
Copper is a transition metal or a d-block element. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
